Portal:Minerals
Portal maintenance status: (May 2019)
|
The Minerals Portal

In geology and mineralogy, a mineral or mineral species is, broadly speaking, a solid substance with a fairly well-defined chemical composition and a specific crystal structure that occurs naturally in pure form.
The geological definition of mineral normally excludes compounds that occur only in living organisms. However, some minerals are often biogenic (such as calcite) or organic compounds in the sense of chemistry (such as mellite). Moreover, living organisms often synthesize inorganic minerals (such as hydroxylapatite) that also occur in rocks.
The concept of mineral is distinct from rock, which is any bulk solid geologic material that is relatively homogeneous at a large enough scale. A rock may consist of one type of mineral or may be an aggregate of two or more different types of minerals, spacially segregated into distinct phases.
Some natural solid substances without a definite crystalline structure, such as opal or obsidian, are more properly called mineraloids. If a chemical compound occurs naturally with different crystal structures, each structure is considered a different mineral species. Thus, for example, quartz and stishovite are two different minerals consisting of the same compound, silicon dioxide. (Full article...)
Mineralogy is a subject of geology specializing in the scientific study of the chemistry, crystal structure, and physical (including optical) properties of minerals and mineralized artifacts. Specific studies within mineralogy include the processes of mineral origin and formation, classification of minerals, their geographical distribution, as well as their utilization. (Full article...)
Selected articles
-
Image 1

Turquoise is an opaque, blue-to-green mineral that is a hydrous phosphate of copper and aluminium, with the chemical formula CuAl6(PO4)4(OH)8·4H2O. It is rare and valuable in finer grades and has been prized as a gemstone for millennia due to its hue.
Like most other opaque gems, turquoise has been devalued by the introduction of treatments, imitations, and synthetics into the market. The robin egg blue or sky blue color of the Persian turquoise mined near the modern city of Nishapur, Iran, has been used as a guiding reference for evaluating turquoise quality. (Full article...) -
Image 2
Opal is a hydrated amorphous form of silica (SiO2·nH2O); its water content may range from 3% to 21% by weight, but is usually between 6% and 10%. Due to its amorphous property, it is classified as a mineraloid, unlike crystalline forms of silica, which are considered minerals. It is deposited at a relatively low temperature and may occur in the fissures of almost any kind of rock, being most commonly found with limonite, sandstone, rhyolite, marl, and basalt.
The name opal is believed to be derived from the Sanskrit word upala (उपल), which means 'jewel', and later the Greek derivative opállios (ὀπάλλιος).
There are two broad classes of opal: precious and common. Precious opal displays play-of-color (iridescence); common opal does not. Play-of-color is defined as "a pseudo chromatic optical effect resulting in flashes of colored light from certain minerals, as they are turned in white light." The internal structure of precious opal causes it to diffract light, resulting in play-of-color. Depending on the conditions in which it formed, opal may be transparent, translucent, or opaque, and the background color may be white, black, or nearly any color of the visual spectrum. Black opal is considered the rarest, while white, gray, and green opals are the most common. (Full article...) -
Image 3
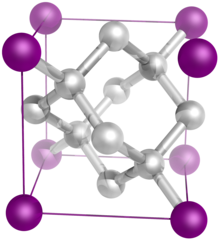
The diamond crystal structure belongs to the face-centered cubic lattice, with a repeated two-atom pattern.
In crystallography, a crystal system is a set of point groups (a group of geometric symmetries with at least one fixed point). A lattice system is a set of Bravais lattices. Space groups are classified into crystal systems according to their point groups, and into lattice systems according to their Bravais lattices. Crystal systems that have space groups assigned to a common lattice system are combined into a crystal family.
The seven crystal systems are triclinic, monoclinic, orthorhombic, tetragonal, trigonal, hexagonal, and cubic. Informally, two crystals are in the same crystal system if they have similar symmetries (though there are many exceptions). (Full article...) -
Image 4
Graphite (/ˈɡræfaɪt/) is a crystalline form of the element carbon. It consists of stacked layers of graphene. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on a large scale (1.3 million metric tons per year in 2022) for uses in pencils, lubricants, and electrodes. Under high pressures and temperatures it converts to diamond. It is a good (but not excellent) conductor of both heat and electricity. (Full article...) -
Image 5Beachy Head is a part of the extensive Southern England Chalk Formation.
Chalk is a soft, white, porous, sedimentary carbonate rock. It is a form of limestone composed of the mineral calcite and originally formed deep under the sea by the compression of microscopic plankton that had settled to the sea floor. Chalk is common throughout Western Europe, where deposits underlie parts of France, and steep cliffs are often seen where they meet the sea in places such as the Dover cliffs on the Kent coast of the English Channel.
Chalk is mined for use in industry, such as for quicklime, bricks and builder's putty, and in agriculture, for raising pH in soils with high acidity. It is also used for "blackboard chalk" for writing and drawing on various types of surfaces, although these can also be manufactured from other carbonate-based minerals, or gypsum. (Full article...) -
Image 6Deep green isolated fluorite crystal resembling a truncated octahedron, set upon a micaceous matrix, from Erongo Mountain, Erongo Region, Namibia (overall size: 50 mm × 27 mm, crystal size: 19 mm wide, 30 g)
Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon.
The Mohs scale of mineral hardness, based on scratch hardness comparison, defines value 4 as fluorite.
Pure fluorite is colourless and transparent, both in visible and ultraviolet light, but impurities usually make it a colorful mineral and the stone has ornamental and lapidary uses. Industrially, fluorite is used as a flux for smelting, and in the production of certain glasses and enamels. The purest grades of fluorite are a source of fluoride for hydrofluoric acid manufacture, which is the intermediate source of most fluorine-containing fine chemicals. Optically clear transparent fluorite has anomalous partial dispersion, that is, its refractive index varies with the wavelength of light in a manner that differs from that of commonly used glases, so fluorite is useful in making apochromatic lenses, making it valuable, particularly in photographic optics. Fluorite optics are also usable in the far-ultraviolet and mid-infrared ranges, where conventional glasses are too opaque for use. Fluorite also has low dispersion, and a high refractive index for its density, which can make it useful for some specialized purposes in optics. (Full article...) -
Image 7

Crystal structure of table salt (sodium in purple, chlorine in green)
In crystallography, crystal structure is a description of the ordered arrangement of atoms, ions, or molecules in a crystalline material. Ordered structures occur from the intrinsic nature of the constituent particles to form symmetric patterns that repeat along the principal directions of three-dimensional space in matter.
The smallest group of particles in the material that constitutes this repeating pattern is the unit cell of the structure. The unit cell completely reflects the symmetry and structure of the entire crystal, which is built up by repetitive translation of the unit cell along its principal axes. The translation vectors define the nodes of the Bravais lattice.
The lengths of the principal axes, or edges, of the unit cell and the angles between them are the lattice constants, also called lattice parameters or cell parameters. The symmetry properties of the crystal are described by the concept of space groups. All possible symmetric arrangements of particles in three-dimensional space may be described by the 230 space groups.
The crystal structure and symmetry play a critical role in determining many physical properties, such as cleavage, electronic band structure, and optical transparency. (Full article...) -
Image 8

Kaolinite (/ˈkeɪ.ələˌnaɪt, -lɪ-/ KAY-ə-lə-nyte, -lih-; also called kaolin) is a clay mineral, with the chemical composition: Al2Si2O5(OH)4. It is a layered silicate mineral, with one tetrahedral sheet of silica (SiO4) linked through oxygen atoms to one octahedral sheet of alumina (AlO6).
Kaolinite is a soft, earthy, usually white, mineral (dioctahedral phyllosilicate clay), produced by the chemical weathering of aluminium silicate minerals like feldspar. It has a low shrink–swell capacity and a low cation-exchange capacity (1–15 meq/100 g).
Rocks that are rich in kaolinite, and halloysite, are known as kaolin (/ˈkeɪ.əlɪn/) or china clay. In many parts of the world kaolin is colored pink-orange-red by iron oxide, giving it a distinct rust hue. Lower concentrations of iron oxide yield the white, yellow, or light orange colors of kaolin. Alternating lighter and darker layers are sometimes found, as at Providence Canyon State Park in Georgia, United States.
Kaolin is an important raw material in many industries and applications. Commercial grades of kaolin are supplied and transported as powder, lumps, semi-dried noodle or slurry. Global production of kaolin in 2021 was estimated to be 45 million tonnes, with a total market value of $US4.24 billion. (Full article...) -
Image 9

Talc, or talcum, is a clay mineral composed of hydrated magnesium silicate, with the chemical formula Mg3Si4O10(OH)2. Talc in powdered form, often combined with corn starch, is used as baby powder. This mineral is used as a thickening agent and lubricant. It is an ingredient in ceramics, paints, and roofing material. It is a main ingredient in many cosmetics. It occurs as foliated to fibrous masses, and in an exceptionally rare crystal form. It has a perfect basal cleavage and an uneven flat fracture, and it is foliated with a two-dimensional platy form.
The Mohs scale of mineral hardness, based on scratch hardness comparison, defines value 1 as the hardness of talc, the softest mineral. When scraped on a streak plate, talc produces a white streak, though this indicator is of little importance, because most silicate minerals produce a white streak. Talc is translucent to opaque, with colors ranging from whitish grey to green with a vitreous and pearly luster. Talc is not soluble in water, and is slightly soluble in dilute mineral acids.
Soapstone is a metamorphic rock composed predominantly of talc. (Full article...) -
Image 10
A rock containing three crystals of pyrite (FeS2). The crystal structure of pyrite is primitive cubic, and this is reflected in the cubic symmetry of its natural crystal facets.
In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals.
There are three main varieties of these crystals:- Primitive cubic (abbreviated cP and alternatively called simple cubic)
- Body-centered cubic (abbreviated cI or bcc)
- Face-centered cubic (abbreviated cF or fcc)
Note: the term fcc is often used in synonym for the cubic close-packed or ccp structure occurring in metals. However, fcc stands for a face-centered-cubic Bravais lattice, which is not necessarily close-packed when a motif is set onto the lattice points. E.g. the diamond and the zincblende lattices are fcc but not close-packed.
Each is subdivided into other variants listed below. Although the unit cells in these crystals are conventionally taken to be cubes, the primitive unit cells often are not. (Full article...) -
Image 11
Diamond is a solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Another solid form of carbon known as graphite is the chemically stable form of carbon at room temperature and pressure, but diamond is metastable and converts to it at a negligible rate under those conditions. Diamond has the highest hardness and thermal conductivity of any natural material, properties that are used in major industrial applications such as cutting and polishing tools. They are also the reason that diamond anvil cells can subject materials to pressures found deep in the Earth.
Because the arrangement of atoms in diamond is extremely rigid, few types of impurity can contaminate it (two exceptions are boron and nitrogen). Small numbers of defects or impurities (about one per million of lattice atoms) can color a diamond blue (boron), yellow (nitrogen), brown (defects), green (radiation exposure), purple, pink, orange, or red. Diamond also has a very high refractive index and a relatively high optical dispersion.
Most natural diamonds have ages between 1 billion and 3.5 billion years. Most were formed at depths between 150 and 250 kilometres (93 and 155 mi) in the Earth's mantle, although a few have come from as deep as 800 kilometres (500 mi). Under high pressure and temperature, carbon-containing fluids dissolved various minerals and replaced them with diamonds. Much more recently (hundreds to tens of million years ago), they were carried to the surface in volcanic eruptions and deposited in igneous rocks known as kimberlites and lamproites.
Synthetic diamonds can be grown from high-purity carbon under high pressures and temperatures or from hydrocarbon gases by chemical vapor deposition (CVD). Imitation diamonds can also be made out of materials such as cubic zirconia and silicon carbide. Natural, synthetic, and imitation diamonds are most commonly distinguished using optical techniques or thermal conductivity measurements. (Full article...) -
Image 12
Magnetite is a mineral and one of the main iron ores, with the chemical formula Fe2+Fe3+2O4. It is one of the oxides of iron, and is ferrimagnetic; it is attracted to a magnet and can be magnetized to become a permanent magnet itself. With the exception of extremely rare native iron deposits, it is the most magnetic of all the naturally occurring minerals on Earth. Naturally magnetized pieces of magnetite, called lodestone, will attract small pieces of iron, which is how ancient peoples first discovered the property of magnetism.
Magnetite is black or brownish-black with a metallic luster, has a Mohs hardness of 5–6 and leaves a black streak. Small grains of magnetite are very common in igneous and metamorphic rocks.
The chemical IUPAC name is iron(II,III) oxide and the common chemical name is ferrous-ferric oxide. (Full article...) -
Image 13Quartz crystal cluster from Brazil
Quartz is a hard, crystalline mineral composed of silica (silicon dioxide). The atoms are linked in a continuous framework of SiO4 silicon–oxygen tetrahedra, with each oxygen being shared between two tetrahedra, giving an overall chemical formula of SiO2. Quartz is, therefore, classified structurally as a framework silicate mineral and compositionally as an oxide mineral. Quartz is the second most abundant mineral in Earth's continental crust, behind feldspar.
Quartz exists in two forms, the normal α-quartz and the high-temperature β-quartz, both of which are chiral. The transformation from α-quartz to β-quartz takes place abruptly at 573 °C (846 K; 1,063 °F). Since the transformation is accompanied by a significant change in volume, it can easily induce microfracturing of ceramics or rocks passing through this temperature threshold.
There are many different varieties of quartz, several of which are classified as gemstones. Since antiquity, varieties of quartz have been the most commonly used minerals in the making of jewelry and hardstone carvings, especially in Europe and Asia.
Quartz is the mineral defining the value of 7 on the Mohs scale of hardness, a qualitative scratch method for determining the hardness of a material to abrasion. (Full article...) -
Image 14Dolomite (white) on talc
Dolomite (/ˈdɒl.əˌmaɪt, ˈdoʊ.lə-/) is an anhydrous carbonate mineral composed of calcium magnesium carbonate, ideally CaMg(CO3)2. The term is also used for a sedimentary carbonate rock composed mostly of the mineral dolomite (see Dolomite (rock)). An alternative name sometimes used for the dolomitic rock type is dolostone. (Full article...) -
Image 15

Corundum is a crystalline form of aluminium oxide (Al2O3) typically containing traces of iron, titanium, vanadium, and chromium. It is a rock-forming mineral. It is a naturally transparent material, but can have different colors depending on the presence of transition metal impurities in its crystalline structure. Corundum has two primary gem varieties: ruby and sapphire. Rubies are red due to the presence of chromium, and sapphires exhibit a range of colors depending on what transition metal is present. A rare type of sapphire, padparadscha sapphire, is pink-orange.
The name "corundum" is derived from the Tamil-Dravidian word kurundam (ruby-sapphire) (appearing in Sanskrit as kuruvinda).
Because of corundum's hardness (pure corundum is defined to have 9.0 on the Mohs scale), it can scratch almost all other minerals. It is commonly used as an abrasive on sandpaper and on large tools used in machining metals, plastics, and wood. Emery, a variety of corundum with no value as a gemstone, is commonly used as an abrasive. It is a black granular form of corundum, in which the mineral is intimately mixed with magnetite, hematite, or hercynite.
In addition to its hardness, corundum has a density of 4.02 g/cm3 (251 lb/cu ft), which is unusually high for a transparent mineral composed of the low-atomic mass elements aluminium and oxygen. (Full article...) -
Image 16

Zeolite exhibited in the Estonian Museum of Natural History
Zeolite is a family of several microporous, crystalline aluminosilicate materials commonly used as commercial adsorbents and catalysts. They mainly consist of silicon, aluminium, oxygen, and have the general formula Mn+
1/n(AlO
2)−
(SiO
2)
x・yH
2O where Mn+
1/n is either a metal ion or H+. These positive ions can be exchanged for others in a contacting electrolyte solution. H+
exchanged zeolites are particularly useful as solid acid catalysts.
The term was originally coined in 1756 by Swedish mineralogist Axel Fredrik Cronstedt, who observed that rapidly heating a material, believed to have been stilbite, produced large amounts of steam from water that had been adsorbed by the material. Based on this, he called the material zeolite, from the Greek ζέω (zéō), meaning "to boil" and λίθος (líthos), meaning "stone".
Zeolites occur naturally, but are also produced industrially on a large scale. , 253 unique zeolite frameworks have been identified, and over 40 naturally occurring zeolite frameworks are known. Every new zeolite structure that is obtained is examined by the International Zeolite Association Structure Commission (IZA-SC) and receives a three-letter designation. (Full article...) -
Image 17

Gypsum is a soft sulfate mineral composed of calcium sulfate dihydrate, with the chemical formula CaSO4·2H2O. It is widely mined and is used as a fertilizer and as the main constituent in many forms of plaster, drywall and blackboard or sidewalk chalk. Gypsum also crystallizes as translucent crystals of selenite. It forms as an evaporite mineral and as a hydration product of anhydrite. The Mohs scale of mineral hardness defines gypsum as hardness value 2 based on scratch hardness comparison.
Fine-grained white or lightly tinted forms of gypsum known as alabaster have been used for sculpture by many cultures including Ancient Egypt, Mesopotamia, Ancient Rome, the Byzantine Empire, and the Nottingham alabasters of Medieval England. (Full article...) -
Image 18
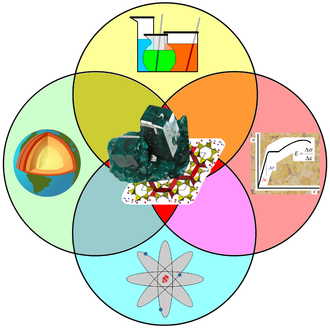
Mineralogy applies principles of chemistry, geology, physics and materials science to the study of minerals
Mineralogy is a subject of geology specializing in the scientific study of the chemistry, crystal structure, and physical (including optical) properties of minerals and mineralized artifacts. Specific studies within mineralogy include the processes of mineral origin and formation, classification of minerals, their geographical distribution, as well as their utilization. (Full article...) -
Image 19

Cinnabar (/ˈsɪnəˌbɑːr/; from Ancient Greek κιννάβαρι (kinnábari)), or cinnabarite (/ˌsɪnəˈbɑːraɪt/), also known as mercurblende is the bright scarlet to brick-red form of mercury(II) sulfide (HgS). It is the most common source ore for refining elemental mercury and is the historic source for the brilliant red or scarlet pigment termed vermilion and associated red mercury pigments.
Cinnabar generally occurs as a vein-filling mineral associated with volcanic activity and alkaline hot springs. The mineral resembles quartz in symmetry and it exhibits birefringence. Cinnabar has a mean refractive index near 3.2, a hardness between 2.0 and 2.5, and a specific gravity of approximately 8.1. The color and properties derive from a structure that is a hexagonal crystalline lattice belonging to the trigonal crystal system, crystals that sometimes exhibit twinning.
Cinnabar has been used for its color since antiquity in the Near East, including as a rouge-type cosmetic, in the New World since the Olmec culture, and in China since as early as the Yangshao culture, where it was used in coloring stoneware.
Associated modern precautions for the use and handling of cinnabar arise from the toxicity of the mercury component, which was recognized as early as ancient Rome. (Full article...) -
Image 20Galena with minor pyrite
Galena, also called lead glance, is the natural mineral form of lead(II) sulfide (PbS). It is the most important ore of lead and an important source of silver.
Galena is one of the most abundant and widely distributed sulfide minerals. It crystallizes in the cubic crystal system often showing octahedral forms. It is often associated with the minerals sphalerite, calcite and fluorite. (Full article...) -
Image 21Malachite from the Democratic Republic of the Congo
Malachite is a copper carbonate hydroxide mineral, with the formula Cu2CO3(OH)2. This opaque, green-banded mineral crystallizes in the monoclinic crystal system, and most often forms botryoidal, fibrous, or stalagmitic masses, in fractures and deep, underground spaces, where the water table and hydrothermal fluids provide the means for chemical precipitation. Individual crystals are rare, but occur as slender to acicular prisms. Pseudomorphs after more tabular or blocky azurite crystals also occur. (Full article...) -
Image 22A ruby crystal from Dodoma Region, Tanzania
Ruby is a pinkish red to blood-red colored gemstone, a variety of the mineral corundum (aluminium oxide). Ruby is one of the most popular traditional jewelry gems and is very durable. Other varieties of gem-quality corundum are called sapphires. Ruby is one of the traditional cardinal gems, alongside amethyst, sapphire, emerald, and diamond. The word ruby comes from ruber, Latin for red. The color of a ruby is due to the element chromium.
Some gemstones that are popularly or historically called rubies, such as the Black Prince's Ruby in the British Imperial State Crown, are actually spinels. These were once known as "Balas rubies".
The quality of a ruby is determined by its color, cut, and clarity, which, along with carat weight, affect its value. The brightest and most valuable shade of red, called blood-red or pigeon blood, commands a large premium over other rubies of similar quality. After color follows clarity: similar to diamonds, a clear stone will command a premium, but a ruby without any needle-like rutile inclusions may indicate that the stone has been treated. Ruby is the traditional birthstone for July and is usually pinker than garnet, although some rhodolite garnets have a similar pinkish hue to most rubies. The world's most valuable ruby to be sold at auction is the Sunrise Ruby. (Full article...) -
Image 23

Tourmaline (/ˈtʊərməlɪn, -ˌliːn/ TOOR-mə-lin, -leen) is a crystalline silicate mineral group in which boron is compounded with elements such as aluminium, iron, magnesium, sodium, lithium, or potassium. This gemstone comes in a wide variety of colors.
The name is derived from the Sinhalese tōramalli (ටෝරමල්ලි), which refers to the carnelian gemstones. (Full article...) -
Image 24

A crystalline solid: atomic resolution image of strontium titanate. Brighter spots are columns of strontium atoms and darker ones are titanium-oxygen columns.
Crystallography is the branch of science devoted to the study of molecular and crystalline structure and properties. The word crystallography is derived from the Ancient Greek word κρύσταλλος (krústallos; "clear ice, rock-crystal"), and γράφειν (gráphein; "to write"). In July 2012, the United Nations recognised the importance of the science of crystallography by proclaiming 2014 the International Year of Crystallography.
Crystallography is a broad topic, and there are many subareas such as X-ray crystallography which are themselves large scientific topics. It spans science ranging from the fundamentals of crystal structure to the mathematics of crystal geometry including those which are not periodic or quasicrystals. At the atomic scale it can involve the use of X-ray diffraction to produce experimental data which the tools of X-ray crystallography can convert into detailed positions of atoms and sometimes electron density. At the larger scale it includes experimental tools such as orientational imaging to examine the relative orientations at the grain boundary in materials. Crystallography plays a key role in many areas of established biology, chemistry and physics as well new developments in these fields. (Full article...) -
Image 25

Rutile is an oxide mineral composed of titanium dioxide (TiO2), the most common natural form of TiO2. Rarer polymorphs of TiO2 are known, including anatase, akaogiite, and brookite.
Rutile has one of the highest refractive indices at visible wavelengths of any known crystal and also exhibits a particularly large birefringence and high dispersion. Owing to these properties, it is useful for the manufacture of certain optical elements, especially polarization optics, for longer visible and infrared wavelengths up to about 4.5 micrometres. Natural rutile may contain up to 10% iron and significant amounts of niobium and tantalum.
Rutile derives its name from the Latin rutilus ('red'), in reference to the deep red color observed in some specimens when viewed by transmitted light. Rutile was first described in 1803 by Abraham Gottlob Werner using specimens obtained in Horcajuelo de la Sierra, Madrid (Spain), which is consequently the type locality. (Full article...)
Selected mineralogist
-
Image 1Ernst Anton Wülfing (27 November 1860, Elberfeld – 17 December 1930, Bad Wörishofen) was a German mineralogist and petrographer, known for his research on the optical properties of minerals and meteorites.
He studied chemistry at Geneva and at Heidelberg as a student of Robert Bunsen, then focused his attention to mineralogy and geology, of which, he studied at Greifswald and Vienna (1887–88). Afterwards he served as an assistant to Harry Rosenbusch at the University of Heidelberg. (Full article...) -
Image 2Charles-Victor Mauguin (French: [ʃaʁl.vik.tɔʁ mo.gɛ̃]; 19 September 1878 – 25 April 1958), more often Charles Mauguin, was a French mineralogist and crystallographer. He and Carl Hermann invented an international standard notation for crystallographic groups called Hermann–Mauguin notation (also sometimes called international notation). (Full article...)
-
Image 3

Maurice Armand Chaper (13 February 1834, Dijon – 5 July 1896, Vienna) was a French geologist and mining engineer.
He received his education at the École Polytechnique and École des Mines, afterwards working at jobs for the railroads and public works. He enlisted in the National Guard during the Franco-Prussian War, rising to the rank of lieutenant-colonel in the 38th Regiment. In 1872 he was named assistant mayor of the 5th arrondissement of Paris. (Full article...) -
Image 4
Evgraf Stepanovich Fedorov (Russian: Евгра́ф Степа́нович Фёдоров, 22 December [O.S. 10 December] 1853 – 21 May 1919) was a Russian mathematician, crystallographer and mineralogist.
Fedorov was born in the Russian city of Orenburg. His father was a topographical engineer. The family later moved to Saint Petersburg. From the age of fifteen, he was deeply interested in the theory of polytopes, which later became his main research interest. He was a distinguished graduate of the Gorny Institute, which he joined at the age of 26. He was elected the first Director of the Institute in 1905. (Full article...) -
Image 5Adolarius Jacob Forster (1739–1806) was a Prussian mineralogist and dealer in display specimen minerals. The Forster family left Yorkshire in 1649 and settled in Prussia. Adolarius Jacob Forster began dealing in mineral specimens around 1766, at the age of 27. He continued in that profession for 40 years and travelled widely. He had premises in London, Paris and St. Petersburg. The Covent Garden, London shop and one in Soho was run by his wife. His brother, Ingham Henry Forster (1725–1782) ran the business in Paris. Auction catalogues for sales in Paris were written by Rome de l'Isle.
He was related to Johann Georg Adam Forster and Johann Reinhold Forster and his sister married the London dealer naturalist George Humphrey at St-Martin-in-the-Fields, London on August 16, 1768. In 1802 Forster sold a collection to the museum of the St Petersburg Mining Institute, under the auspices of the Emperor of All Russia Alexander I. He spent the last ten years of his life in Russia, and died in St. Petersburg in 1806. The dealership was taken over by his nephew John Henry Heuland. (Full article...) -
Image 6
Linus Carl Pauling FRS (/ˈpɔːlɪŋ/ PAW-ling; February 28, 1901 – August 19, 1994) was an American chemist, biochemist, chemical engineer, peace activist, author, and educator. He published more than 1,200 papers and books, of which about 850 dealt with scientific topics. New Scientist called him one of the 20 greatest scientists of all time. For his scientific work, Pauling was awarded the Nobel Prize in Chemistry in 1954. For his peace activism, he was awarded the Nobel Peace Prize in 1962. He is one of five people to have won more than one Nobel Prize (the others being Marie Curie, John Bardeen, Frederick Sanger, and Karl Barry Sharpless). Of these, he is the only person to have been awarded two unshared Nobel Prizes, and one of two people to be awarded Nobel Prizes in different fields, the other being Marie Curie.
Pauling was one of the founders of the fields of quantum chemistry and molecular biology. His contributions to the theory of the chemical bond include the concept of orbital hybridisation and the first accurate scale of electronegativities of the elements. Pauling also worked on the structures of biological molecules, and showed the importance of the alpha helix and beta sheet in protein secondary structure. Pauling's approach combined methods and results from X-ray crystallography, molecular model building, and quantum chemistry. His discoveries inspired the work of Rosalind Franklin, James Watson, Francis Crick, and Maurice Wilkins on the structure of DNA, which in turn made it possible for geneticists to crack the DNA code of all organisms. (Full article...) -
Image 7Helen Dick Megaw (1 June 1907 – 26 February 2002) was an Irish crystallographer who was a pioneer in X-ray crystallography. She made measurements of the cell dimensions of ice and established the Perovskite crystal structure. (Full article...)
-
Image 8

George Washington Carpenter (July 31, 1802 – June 7, 1860) was an American scientist. (Full article...) -
Image 9Philip James Yorke (13 October 1799 – 14 December 1874) was a British Army officer, scientist and Fellow of the Royal Society. Yorke was a founder of the Chemical Society in the United Kingdom. (Full article...)
-
Image 10Paul Heinrich von Groth (1910); photograph by Theodor Hilsdorf
Paul Heinrich Ritter von Groth (23 June 1843 – 2 December 1927) was a German mineralogist. His most important contribution to science was his systematic classification of minerals based on their chemical compositions and crystal structures. (Full article...) -
Image 11Karl Ludwig Felix Machatschki (22 September 1895 – 17 February 1970) was an Austrian mineralogist.
He was born in Arnfels (near Leibnitz) in Styria, Austria. He studied at the University of Graz, obtaining his habilitation in 1925; in 1927 he joined the group of Victor Goldschmidt in Oslo for one year. In 1930 he was appointed as a professor at the University of Tübingen. He changed university twice, first in 1941 to the University of Munich and finally in 1944 to the University of Vienna. (Full article...) -
Image 12

Franz Xaver von Wulfen
Franz Xaver Freiherr von Wulfen (5 November 1728 – 17 March 1805) was an Austrian botanist, zoologist, mineralogist, alpinist, and Jesuit priest. He is credited with discovering the flowering plants Wulfenia carinthiaca, Saxifraga moschata, and Stellaria bulbosa. In 1845 the lead molybdate mineral wulfenite was named in his honor by Wilhelm Karl von Haidinger. (Full article...) -
Image 13Sarah Mawe (1767-1846) was an English mineralogist of the 19th century, appointed to serve Queen Victoria in that capacity from 1837 to 1840. (Full article...)
-
Image 14

William Phipps Blake (June 1, 1826 – May 22, 1910) was an American geologist, mining consultant, and educator. Among his best known contributions include being the first college trained chemist to work full-time for a United States chemical manufacturer (1850), and serving as a geologist with the Pacific Railroad Survey of the Far West (1853–1856), where he observed and detailed a theory on erosion by wind-blown sand on the geologic formations of southern California, one of his many scientific contributions. He started several western mining enterprises that were premature, including a mining magazine in the 1850s and the first school of mines in the Far West in 1864.
From the 1850s on he published over 200 articles, several books, and numerous newspaper and mining magazine columns or short pieces on mining and geology. He served throughout his long career as a mining consultant for mining corporations in every western state and several foreign countries, including Japan. He also served as special ambassador for the nascent science of geology while serving as the United States' principal geologic exhibit commissioner for what now would be called World Fairs, from Paris in 1867, through Vienna and the centennial at Philadelphia, back to Paris in 1878. He ended his long and distinguished career as head of the school of mines at the University of Arizona, 1895–1905, remaining in an active emeritus status until his death. (Full article...) -
Image 15
Prof William Hallowes Miller FRS HFRSE LLD DCL (6 April 1801 – 20 May 1880) was a Welsh mineralogist and laid the foundations of modern crystallography.
Miller indices are named after him, the method having been described in his Treatise on Crystallography (1839). The mineral known as millerite is named after him. (Full article...) -
Image 16
Sten Anders Hjalmar Sjögren (13 June 1856, Färnebo, Värmland – 23 March 1922, Stockholm) was a Swedish geologist and mineralogist. (Full article...) -
Image 17Jean-André Mongez (21 November 1750 – May 1788) was a French priest and mineralogist. He is presumed to have died at Vanikoro, on the La Pérouse expedition. (Full article...)
-
Image 18

William Phillips
William Phillips FGS FRS (10 May 1775 – 2 April 1828) was an English mineralogist and geologist. (Full article...) -
Image 19

portrait of Oskar Lenz
Oskar Lenz (13 April 1848 in Leipzig – 1 March 1925 in Sooß) was a German-Austrian geologist and mineralogist born in Leipzig. (Full article...) -
Image 20

Prof Ferdinand Zirkel FRS(For) HFRSE (20 May 1838 – 11 June 1912) was a German geologist and petrographer. (Full article...) -
Image 21

Johann Friedrich Ludwig Hausmann
Johann Friedrich Ludwig Hausmann (22 February 1782, Hannover – 26 December 1859, Göttingen) was a German mineralogist. (Full article...) -
Image 22
Karl Georg von Raumer (9 April 1783 – 2 June 1865) was a German geologist and educator. (Full article...) -
Image 23
Vladimir Ivanovich Vernadsky, also spelt Volodymyr Ivanovych Vernadsky (Russian: Владимир Иванович Вернадский, Ukrainian: Володимир Іванович Вернадський; 12 March [O.S. 28 February] 1863 – 6 January 1945) was a Russian, Ukrainian, and Soviet mineralogist and geochemist who is considered one of the founders of geochemistry, biogeochemistry, and radiogeology. He was one of the founders and the first president of the Ukrainian Academy of Sciences (now National Academy of Sciences of Ukraine). Vladimir Vernadsky is most noted for his 1926 book The Biosphere in which he inadvertently worked to popularize Eduard Suess' 1875 term biosphere, by hypothesizing that life is the geological force that shapes the earth. In 1943 he was awarded the Stalin Prize. Vernadsky's portrait is depicted on the Ukrainian ₴1,000 hryvnia banknote. (Full article...) -
Image 24Alexander Sadebeck (26 June 1843 in Breslau – 9 December 1879 in Hamburg) was a German geologist and mineralogist. He was a brother of botanist Richard Sadebeck (1839–1905).
He studied mineralogy and geology at the University of Berlin as a pupil of Gustav Rose. In 1865 he received his doctorate with a dissertation on Upper Jurassic formations in Pomerania. In 1872 he was appointed professor of mineralogy and geology at the University of Kiel. (Full article...) -
Image 25

Wolfgang Sartorius von Waltershausen drawn in May 22nd 1843 in Rome by August Kestner
Wolfgang Sartorius Freiherr von Waltershausen (17 December 1809 – 16 March 1876) was a German geologist. (Full article...)
Related portals
Get involved
For editor resources and to collaborate with other editors on improving Wikipedia's Minerals-related articles, see WikiProject Rocks and minerals.
General images
-
Image 1Diamond is the hardest natural material, and has a Mohs hardness of 10. (from Mineral)
-
Image 3Native gold. Rare specimen of stout crystals growing off of a central stalk, size 3.7 x 1.1 x 0.4 cm, from Venezuela. (from Mineral)
-
Image 5When minerals react, the products will sometimes assume the shape of the reagent; the product mineral is termed a pseudomorph of (or after) the reagent. Illustrated here is a pseudomorph of kaolinite after orthoclase. Here, the pseudomorph preserved the Carlsbad twinning common in orthoclase. (from Mineral)
-
Image 7Perfect basal cleavage as seen in biotite (black), and good cleavage seen in the matrix (pink orthoclase). (from Mineral)
-
Image 8Muscovite, a mineral species in the mica group, within the phyllosilicate subclass (from Mineral)
-
Image 10Pink cubic halite (NaCl; halide class) crystals on a nahcolite matrix (NaHCO3; a carbonate, and mineral form of sodium bicarbonate, used as baking soda). (from Mineral)
-
Image 11Mohs hardness kit, containing one specimen of each mineral on the ten-point hardness scale (from Mohs scale)
-
Image 12Sphalerite crystal partially encased in calcite from the Devonian Milwaukee Formation of Wisconsin (from Mineral)
-
Image 13Mohs Scale versus Absolute Hardness (from Mineral)
-
Image 14Asbestiform tremolite, part of the amphibole group in the inosilicate subclass (from Mineral)
-
Image 16Epidote often has a distinctive pistachio-green colour. (from Mineral)
-
Image 20Black andradite, an end-member of the orthosilicate garnet group. (from Mineral)
-
Image 21Red cinnabar (HgS), a mercury ore, on dolomite. (from Mineral)
-
Image 22Schist is a metamorphic rock characterized by an abundance of platy minerals. In this example, the rock has prominent sillimanite porphyroblasts as large as 3 cm (1.2 in). (from Mineral)
-
Image 23Hübnerite, the manganese-rich end-member of the wolframite series, with minor quartz in the background (from Mineral)
-
Image 25An example of elbaite, a species of tourmaline, with distinctive colour banding. (from Mineral)
-
Image 26Gypsum desert rose (from Mineral)
In the news
- 3 May 2024 –
- Panama bans First Quantum Minerals from extracting copper following the closure of its Cobre Panamá mine last year. (Reuters) (The Globe and Mail)
Did you know ...?
- ... that while manganese-bearing vesuvianite has been studied since 1883, manganvesuvianite (pictured) was not described until 2002?
- ... that the mineral rapidcreekite was discovered in Yukon, Canada, in 1983?
- ... that collinsite was discovered in British Columbia and named for the director of the Geological Survey of Canada?
Subcategories
Topics
| Overview | ||
|---|---|---|
| Common minerals | ||
Ore minerals, mineral mixtures and ore deposits | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ores |
| ||||||||
| Deposit types | |||||||||
| Borates | |||||
|---|---|---|---|---|---|
| Carbonates | |||||
| Oxides |
| ||||
| Phosphates | |||||
| Silicates | |||||
| Sulfides | |||||
| Other |
| ||||
| Crystalline | |||||||
|---|---|---|---|---|---|---|---|
| Cryptocrystalline | |||||||
| Amorphous | |||||||
| Miscellaneous | |||||||
| Notable varieties |
| ||||||
| Oxide minerals |
| ||||
|---|---|---|---|---|---|
| Silicate minerals | |||||
| Other | |||||
Gemmological classifications by E. Ya. Kievlenko (1980), updated | |||||||||
| Jewelry stones |
| ||||||||
| Jewelry-Industrial stones |
| ||||||||
| Industrial stones |
| ||||||||
Mineral identification | |
|---|---|
| "Special cases" ("native elements and organic minerals") |
|
|---|---|
| "Sulfides and oxides" |
|
| "Evaporites and similars" |
|
| "Mineral structures with tetrahedral units" (sulfate anion, phosphate anion, silicon, etc.) |
|
Associated Wikimedia
The following Wikimedia Foundation sister projects provide more on this subject:
-
Commons
Free media repository -
Wikibooks
Free textbooks and manuals -
Wikidata
Free knowledge base -
Wikinews
Free-content news -
Wikiquote
Collection of quotations -
Wikisource
Free-content library -
Wikiversity
Free learning tools -
Wiktionary
Dictionary and thesaurus
References
- Pages with French IPA
- Manually maintained portal pages from May 2019
- All manually maintained portal pages
- Portals with triaged subpages from May 2019
- All portals with triaged subpages
- Portals with named maintainer
- Automated article-slideshow portals with 31–40 articles in article list
- Automated article-slideshow portals with 201–500 articles in article list
- Portals needing placement of incoming links